
24 Feb From Constraint to Care: How Frugal Medical Devices Expand Access Across Health Systems
For decades, medical innovation has been defined by technological sophistication such as advanced imaging, robotic precision, and specialist-dependent diagnostics designed for infrastructure-rich health systems. While these advances have improved outcomes in high-income markets, they have also reinforced a structural mismatch between global health needs and the technologies designed to serve them.
As of 2026, a counter-trend is gaining a strategic momentum. Across low- and middle-income countries (LMICs), innovators are redesigning medical devices around constraint rather than abundance. The result is a growing class of frugal medical technologies that deliver clinically meaningful outcomes without relying on stable electricity, specialist labour, or fragile supply chains.
For global health systems, these devices are not marginal substitutes. They represent a scalable blueprint for resilient care delivery, increasingly outperforming legacy models on social return on investment (SROI).
Closing the Infrastructure Alignment Gap
Table 1: The Resource Divergence
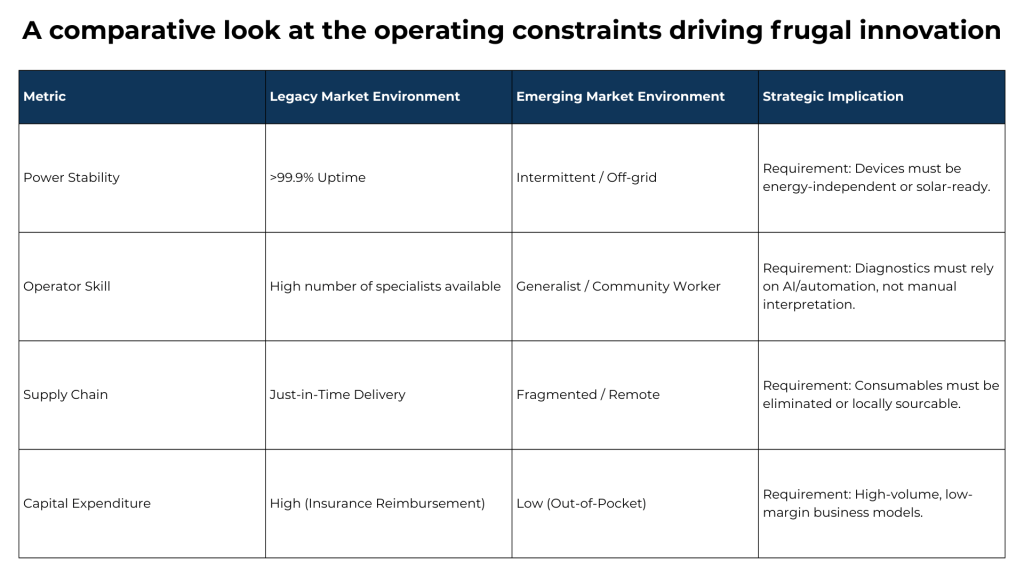
Source: World Health Organization (WHO) & World Bank, Medical & Biological Engineering & Computing Journal; WHO Medical Device Donations Guidelines, World Bank Open Data & WHO Global Health Expenditure Database.
These constraints are not barriers to innovation; they are its primary design inputs.
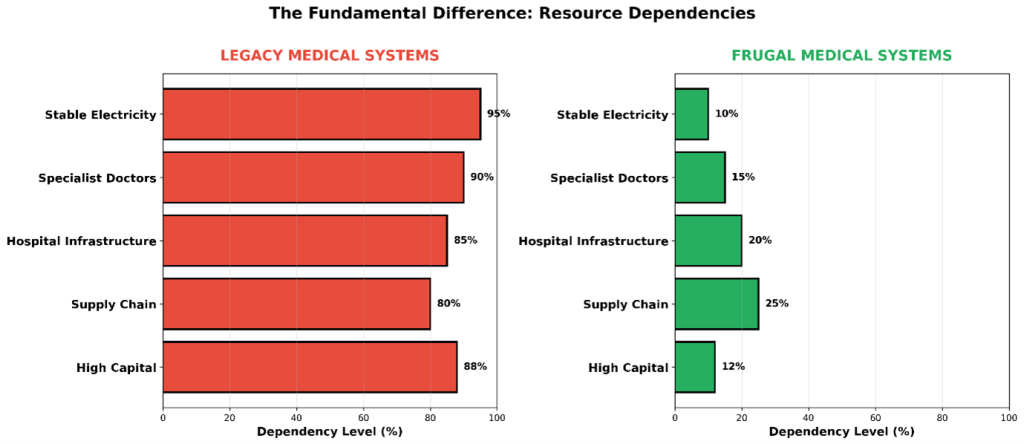
Key Challenges to Widespread Adoption: Policy and Market Implications
Scaling frugal medical devices from pilot initiatives to national health system standards is less a question of technological readiness and more a question of systemic alignment. While many frugal innovations have demonstrated clinical efficacy and cost efficiency, their adoption at scale remains constrained by policy, financing, and market structures originally designed for capital-intensive, hospital-centric technologies. Three structural challenges are particularly significant.
- Regulatory Misalignment
Most regulatory frameworks do not sufficiently differentiate between high-risk, capital-intensive medical equipment and low-risk, point-of-care or screening devices. As a result, frugal medical technologies are often subject to lengthy approval timelines, high regulatory fees, and evidence requirements that are disproportionate to their risk profile. This slows market entry, increases costs for innovators, and limits the ability of health systems to rapidly deploy solutions aligned with urgent public health priorities such as maternal health, early cancer detection, and disability screening.
- Treatment-Heavy Health Financing Models
National health budgets remain predominantly oriented toward tertiary and curative care, with preventive and screening interventions often treated as discretionary or project-based expenditures. This bias towards treatment over prevention limits sustained demand for frugal devices, despite evidence that early detection and decentralised care reduce long-term system costs. Without dedicated financing mechanisms, many high-impact technologies remain confined to donor-funded pilots or small-scale deployments.
- Fragmented Manufacturing and Service Ecosystems
Frugal medical devices derive much of their value from low total cost of ownership and long operational lifespans. However, high import tariffs on components, reliance on centralised manufacturing, and limited local repair capacity undermine these advantages. In many markets, devices become unusable not because of technological failure, but due to delayed maintenance, unavailable spare parts, or unaffordable replacement costs.
How Frugal Innovation Eliminates Barriers Across Care Pathways
Table 2: Device-Led Innovation in Practice
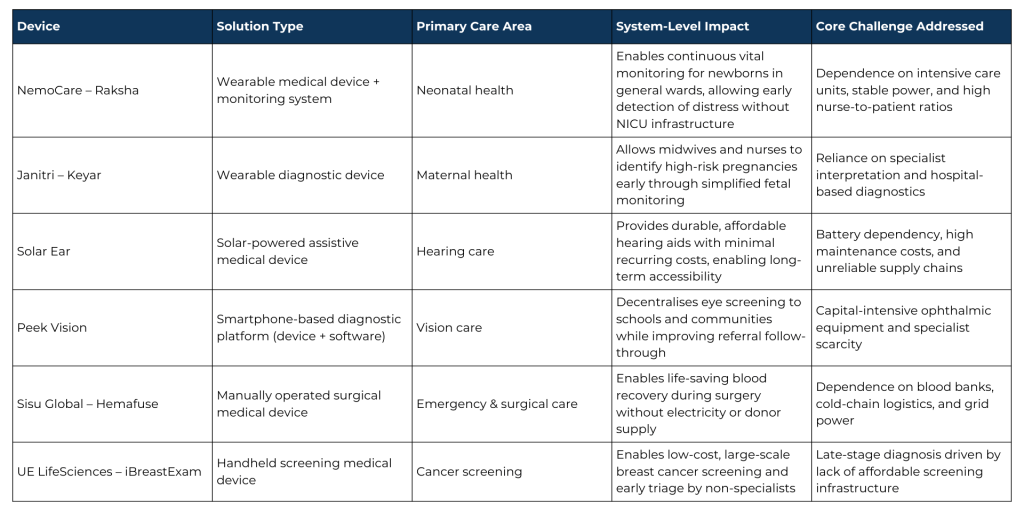
Maternal and Neonatal Health:
Neonatal and fetal monitoring has traditionally been tied to intensive care infrastructure, limiting access in public hospitals and rural settings. Two Indian-origin devices illustrate how frugal engineering is reshaping this model.
Maternal and Neonatal Health: Decentralising Critical Monitoring
NemoCare – Raksha
Raksha is a lightweight wearable sensor placed on a newborn’s foot that continuously measures SpO₂, heart rate, and body temperature. The device transmits data over a local network to a central monitor without requiring constant internet connectivity or grid power. Automated alerts flag early signs of distress, enabling timely intervention by nursing staff. In practice, Raksha has been deployed across government and private hospitals in India, monitoring tens of thousands of newborns in states including Telangana, Karnataka, Tamil Nadu, Maharashtra, and Punjab, helping expand high-quality neonatal surveillance outside traditional NICUs.
Impact: Early deterioration detection moves from NICU dependency to general wards, expanding surveillance without expanding infrastructure.
Janitri – Keyar
Keyar is a wearable fetal monitoring patch that captures fetal heart rate and uterine activity during antenatal care. Embedded algorithms interpret physiological signals and convert them into simple colour-coded alerts, removing the need for real-time specialist interpretation. The device is designed for routine use by midwives and nurses in low-resource settings. Janitri’s solutions are deployed in over 27 Indian states across public and private health facilities and are used for continuous maternal and fetal monitoring during pregnancy and labor, with broader use in home-based care contexts as well.
Impact: Risk identification occurs earlier and closer to the patient, reducing emergency referrals and maternal complications.
Sensory Accessibility:
Solar Ear
Solar Ear is a behind-the-ear hearing aid powered by rechargeable batteries that are charged using a compact solar charger. By eliminating disposable batteries, the device reduces recurring costs and dependence on unreliable supply chains. Local assembly and repair models further extend device lifespan and accessibility. Solar Ear products are used in multiple countries across Africa, Asia, and Latin America, often in community-based hearing programs where battery access is limited.
Impact: Hearing care shifts from a recurring financial burden to a durable, community-sustained service.
Peek Vision
Peek Vision combines a smartphone-based retinal imaging adapter with a software platform that guides non-specialists through eye screening. The system captures images, assesses risk, and manages digital referrals, ensuring patients who need care are efficiently linked to healthcare service providers. Peek-powered programmes have screened over 18 million people across more than a dozen countries in Africa and Asia, connecting more than 1 million individuals to eye care services and supporting school and community eye health initiatives.
Impact: Screening is decentralised to schools and communities while preserving specialist capacity for confirmed cases.
Surgical Resilience:
Hemafuse by Sisu Global
Hemafuse is a handheld, manually operated autotransfusion device used during internal bleeding. It collects, filters, and reinfuses a patient’s own blood in real time, without electricity or donor blood. The purely mechanical design ensures reliability in emergency and off-grid surgical settings. Hemafuse is currently used in emergency and surgical settings in countries like Ghana and Kenya, helping address critical blood shortages and reducing dependence on donor blood.
Impact: Surgical outcomes improve as dependence on external blood supply and cold-chain logistics is removed.
Early Detection at Scale:
iBreastExam by UE LifeSciences (Asia / Latin America)
iBreastExam is a handheld, radiation-free breast screening device that uses tactile sensors to detect variations in tissue elasticity. Operated by trained health workers, it provides immediate results and flags cases requiring further diagnostic follow-up. The device has been deployed across health programmes in Asia and Latin America, enabling early triage for breast cancer among underserved populations who otherwise lack access to mammography.
Impact: Cancer detection shifts from late-stage diagnosis to early outpatient triage, lowering treatment costs and preserving household economic stability.
Recommendations for Scaling Frugal Medical Devices
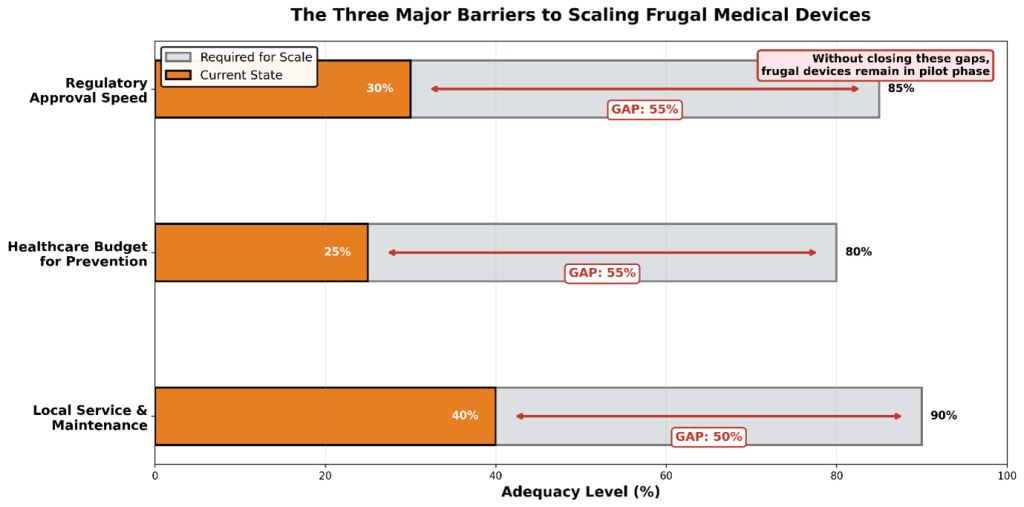
Experience across multiple health systems suggests that frugal innovations scale most effectively when policy and market structures evolve alongside technology deployment. Several best practices are emerging across regions.
- Proportionate and Tiered Regulatory Pathways
Health authorities that differentiate regulatory requirements based on device risk and intended use are better positioned to integrate frugal technologies at scale. Tiered approval pathways particularly for WHO-prequalified, low-risk screening and monitoring devices can reduce time-to-market while maintaining patient safety. Proportionate regulation enables health systems to respond more quickly to high-burden health challenges without compromising oversight.
- Embedding Prevention into Core Health Financing
Successful scaling of frugal devices is closely linked to predictable, budget-backed demand. Health systems that formally allocate funding for preventive screening and early detection especially within maternal health, non-communicable disease, and disability programmes, create stable procurement pathways for these technologies. Shifting even a modest share of expenditure from late-stage treatment toward early intervention has been shown to improve outcomes while containing long-term costs.
- Strengthening Local Assembly and Maintenance Capacity
Markets that support local assembly, repair, and servicing of medical devices consistently achieve higher uptime and lower lifecycle costs. Reducing tariffs on components, supporting technician training, and encouraging procurement models that value serviceability over one-time purchase price all contribute to more resilient device ecosystems. Localised supply chains not only improve sustainability but also enhance system responsiveness during disruptions.
Conclusion
The emergence of frugal medical devices signals a structural shift in how healthcare innovation is being conceived and deployed. Across maternal and neonatal monitoring, sensory accessibility, surgical care, and early cancer detection, the devices examined in this article demonstrate that high clinical impact no longer depends on complex infrastructure or specialist density. Instead, effectiveness is increasingly driven by designs that align with real-world constraints such as unreliable power, limited workforce capacity, and fragmented supply chains, while delivering essential functionality at scale.
A clear pattern underpins these successes. Devices that reduce dependency on grid power, specialist interpretation, and recurring consumables enable earlier intervention and broader access, shifting care pathways from late-stage treatment to prevention, screening, and decentralised delivery. This transition lowers total cost of ownership, improves system resilience, and generates higher social return on investment, particularly in health systems under fiscal and operational pressure.
Yet technology alone is insufficient. Without proportionate regulation, financing models that prioritise prevention, and ecosystems that support local assembly and maintenance, frugal innovations remain confined to pilots rather than becoming national standards. Where these best practices are in place, frugal medical devices evolve from stop-gap solutions into foundational health infrastructure. As global health systems confront rising costs and access gaps, the central question is no longer whether frugal innovation works, but whether health systems are ready to adapt to the model it enables.




